PongMoji/Shutterstock.com
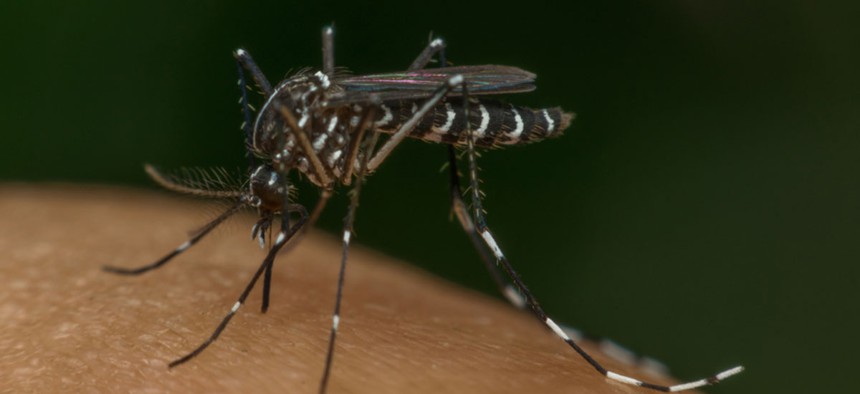
Screening for Zika in Donated Blood
The Food and Drug Administration now recommends that all blood banks test for the mosquito-borne virus.
The U.S. government is now recommending that blood banks around the nation take precautions against the Zika virus.
The Food and Drug Administration announced Friday it will recommend all U.S. blood banks screen donated blood for the mosquito-borne virus, which can cause birth defects. In February, the FDA recommended screening only in areas with confirmed Zika cases, as well as barring donations from people who had recently visited such areas or had been exposed.
Noting the general uncertainty surrounding Zika, and the fact that four out of five infected people don’t show symptoms of the virus, Dr. Luciana Borio, the FDA’s acting chief scientist, said in a statement:
As new scientific and epidemiological information regarding Zika virus has become available, it’s clear that additional precautionary measures are necessary. We are issuing revised guidance for immediate implementation in order to help maintain the safety of the U.S. blood supply.
The FDA said that in light of the new guidance, it will test already-donated blood in Florida and Puerto Rico.
As scientists study the virus—and how it can effectively spread through sexual intercourse—Zika cases continue to pop up in Florida, Puerto Rico, and the U.S. Virgin Islands, along with South America and the Caribbean. On Tuesday, Florida officials said they were investigating five Zika cases where the virus was transmitted locally. Around 50 people have now contracted the virus in South Florida.






